Metsera is a clinical-stage biopharmaceutical company accelerating the next generation of medicines for obesity and metabolic diseases. Metsera is advancing a broad portfolio of oral and injectable incretin, non-incretin and combination therapies with potential best-in-class profiles to address multiple therapeutic targets and meet the future needs of a rapidly evolving weight loss treatment landscape. Metsera went public in 2025 (NASDAQ: MTSR). The company was subsequently acquired by Pfizer in 2025 and is no longer publicly traded.
Sector: Life Sciences
nChromaBio
nChroma Bio is a pioneering biotechnology company redefining the future of in vivo targeted genetic medicine to treat a wide array of diseases and bring cures to patients. The company’s integrated product engine tackles significant limitations of existing genetic medicine approaches by enabling safe, precise and specific in vivo delivery. nChroma’s near clinical-stage development candidate, CRMA-1001, is a liver-targeted therapy in development as a potential functional cure for chronic hepatitis B and hepatitis D that leverages the power of epigenetics, nature’s innate mechanism for gene regulation. Guided by a world-class team at the forefront of genetic medicine, founded by renowned pioneers in the field, and supported by top-tier investors, the company is uniquely positioned to deliver groundbreaking therapies with programmable tissue specificity, unlocking highly potent, durable and targeted gene regulation for the liver and beyond.
Engrail Therapeutics
Engrail Therapeutics is a clinical-stage pharmaceutical company with the aspiration of becoming a leader in neuroscience. Their purpose is to deliver transformational therapies that improve the lives of patients with neuropsychiatric and neurodevelopmental diseases.
EurekaBio
Through the synergies of cross-disciplinary collaboration and technological innovation, EurekaBio has consistently pushed the boundaries of therapy. EurekaBio has brought forth advanced cellular drug production solutions and the EuLV™ lentiviral vector platform. These breakthroughs, including high-titer stable lentiviral vector packaging and producer cell lines, are poised to play a pivotal role in the global commercialization of CGT drugs. Our ultimate aim is to bring tangible benefits to patients worldwide by ushering in a new era of advanced and accessible therapies.
Behind the Breakthrough: Q&A with Peptone, Kamil Tamiola
Kamil Tamiola’s approach to better understand disease-causing proteins reflects his passion to study protein disorders.
Intrinsically disordered proteins (dubbed IDPs) and intrinsically disordered regions (IDRs) occur in one-third of the human proteome. Disordered proteins are shape-shifting and lack a consistent 3D structure. As a result, IDPs cannot be visualized using existing experimental methods, such as microscopy and mass spectrometry, or by predictive modeling approaches. Determined to “see” the invisible in the disordered proteome, Tamiola and his team have used interdisciplinary collaboration to address this longstanding challenge.
Tamiola had an opportunity early in his career to collaborate with researchers studying proteins implicated in progressive neurodegenerative disorders, like human alpha-synuclein and tau.
He explained, “It became evident that my expertise alone could only make a limited impact, and the true excitement lay in bringing together multiple disciplines.” The interdisciplinary collaboration was a significant shift for Tamiola, transitioning from solo physicist to cross-functional collaborator working closely with biologists and protein engineers. It was through this blending of different scientific disciplines that Peptone was born.
Peptone’s approach combines experimental and computational methods to study IDP molecular motions and their implications in biology. In a competitive landscape where some companies are using traditional approaches to tackle these challenging targets, Tamiola believes “Peptone’s technology can redefine what is considered “undruggable” into potential therapeutic opportunities.”
By leveraging proprietary tools to study protein shape and behavior before pursuing binding studies, Peptone can build more reliable models for drug discovery. This parallels the shift that X-ray crystallography brought to folded proteins, where understanding structure led to computer-aided molecule design.
What motivated you to start Peptone?
The idea stemmed from realizing the underexplored potential of IDPs while collaborating in academia. Historically, drug discovery against disordered proteins involved screening, identifying binding molecules and then refining them through tools like NMR spectroscopy. With a bit of luck, we recognized how little effort had been devoted to this incredibly important class of proteins and set out to develop drug candidates that would bind to these proteins and exert a biological effect.
We began with some thrilling results that came out of proof-of-concept studies with pharmaceutical partners. We demonstrated the transformative power of using computers to alter the properties of disordered proteins and yield empirical results, all achieved without the existence of our own laboratory at the time.
These successful early endeavors motivated us to establish a dedicated laboratory, where we now engage in truly original research, and that was the moment that marked the inception of Peptone.
“It became evident that my expertise alone could only make a limited impact, and the true excitement lay in bringing together multiple disciplines.”
Dr. Kamil Tamiola, Founder & CEO, Peptone
Why is understanding the structure of intrinsically disordered proteins so important for advancing human health?
Intrinsically disordered proteins are widespread in the human proteome and across all life forms. They serve the purpose of enabling proteins to exhibit diverse, dynamic, and plastic structures to fulfill complex cellular functions. Unlike proteins with a singular function, these proteins collaborate with partners and can assume various structures. Considering the role of IDPs in numerous biological functions, both inside and outside the cell, there is a vast array of potential therapeutic targets.
IDPs are a large and diverse group and early research focused on neurodegeneration. This interest arose from the observation that bizarre, yet structured fibers found in human brains caused Alzheimer’s. Notably, the molecules forming these fibers lacked any inherent structure themselves.
Today, we recognize that in oncology, cardiology, autoimmune diseases, and even weight loss, disordered peptides play a crucial role – highlighting the broad relevance of disordered proteins across various therapeutic categories.
What makes you most hopeful about Peptone’s future?
We are seeing incredibly exciting data coming out of our programs and it is abundantly clear to us that this technology that we first developed for biologics research can also be successfully applied to small molecule drug discovery for intracellular targets.
As you read this, we are generating a wealth of data on challenging disordered targets showing great promise with clear therapeutic hypotheses. Our innovative insights position us to create best-in-class or first-in-class solutions. This is incredibly exciting as it allows us to revisit areas where people acknowledged the importance of a target but lacked a starting point. With Peptone’s unique approach and supporting data, we can now confidently guide progress.
How do think about the impact your early research can have on humankind overall?
I’m a humanist, and I believe in the value of humankind. And I still believe even with AI advances there will be a space and place and purpose for us in this world. Most importantly, I hope that companies like us, irrespective of how prolific we’ll be, will move the needle towards a better understanding of all these debilitating diseases and to giving people a better life.
What’s one lesson you’ve learned so far as CEO and founder?
Drug discovery is incredibly difficult with so many things that can go wrong. As much as we wish that we could reduce it to engineering and technological problems, I just don’t believe in that. What is incredibly humbling is the tremendous amount of artistry, devotion, and drive that individuals in the company must have, especially when there’s a challenge.
It is important to have a team that is motivated, almost obsessed with details, and in the face of setbacks, can find the inspiration to repeat experiments, look once again over data, and push in advance. So as CEO, my biggest responsibility is to communicate the progress of ups and downs transparently and concisely so we can make sensible decisions.
Who is someone who’s had the most professional impact on your career, and why?
I have two mentors. One is Dr. George Golumbeski and the other is Dr. Andrew Allen, both members of Peptone’s board of directors. Golumbeski is known in the drug discovery field as one of the most prolific deal makers who was key in the acquisition of Celgene by BMS. Andrew Allen is a professional chairman of our company but also an entrepreneur and clinician himself. They are both incredible people and play a vital role in shaping how I run the company and how I think about drug discovery. I call them my sounding board when it comes to decision-making, but also strategic thinking. Sometimes it is not even by giving me direct feedback, but by sending an article or a podcast saying, “Listen to this, look like these guys did it, what is there for us?” So, providing a learning experience in a variety of ways and I’m very, very grateful for that.
What’s one regular habit of yours that’s integral to your professional happiness and success?
Daily grand piano practice and reflection are very very useful. They really help me to calm down and focus and allow me to put myself in a sort of sensory deprivation mode to counteract all the stimuli I am exposed to daily. I’m also very committed to getting seven to eight hours of sleep every night and that is one thing I would recommend to anybody.
Subscribe to our newsletter to get the latest updates on how our portfolio companies continue to lead the way in their respective fields.
OnCusp Therapeutics
OnCusp Therapeutics, Inc., headquartered in New York City, is a biopharmaceutical company dedicated to transforming cutting-edge preclinical innovation into clinically validated treatments for cancer patients worldwide. OnCusp was co-founded by Dr. Bing Yuan, Dr. Eric Slosberg, and Dr. Andy Fu, and has built a strong team of accomplished veterans with proven track records in building startup biotechs, leading successful preclinical and clinical programs, and creating value through global partnerships. The company is committed to accelerating the advancement of globally competitive oncology assets for patients.
Providing the next generation of high-impact medicines
Biologics and peptides have revolutionized healthcare setting the stage for a new generation of equally transformative orally available small molecule medicines.
A critical gap in understanding GPCR biology has long held back an entire therapeutic category
G-protein coupled receptors (GPCRs) – a family of receptors that sense and respond to external signals – comprise the largest family of targets for approved drugs with 826 identified members, which represents approximately 475 drugs on the market acting on over 100 unique GPCRs. Abnormal GPCR activity has been directly linked to various chronic diseases including cardiovascular, metabolic, and pulmonary conditions. Despite the wealth of commercialized compounds, these proteins remain one of the most challenging target classes for structure-based drug discovery because advances in our overall knowledge of receptor structure and function have been relatively slow. As a result, more than 220 GPCRs have not yet been explored as clinical targets.
“Because the structure and function of biological macromolecules (i.e., proteins) are intimately intertwined, structural biology-based approaches offer an important foundation for drug discovery,” said Raymond Stevens, PhD, Founder and CEO of Structure Therapeutics. “Researchers have long sought out structural insights to gain intel about some of the most challenging targets (including GPCRs) across disease indications, and F-Prime shares in our commitment to developing differentiated small molecule therapies to overcome the limitations of biologics and peptide therapies and provide better treatment options for patients everywhere.”
Small molecules are a category of medicine that offers substantial benefits such as their oral administration and design, which optimizes and targets distribution in the body. In addition, their potential for patient compliance is higher and they offer more flexibility than biologics.
One small discovery could pave the way to one large leap in medicine
Structure Therapeutics’ story began several years ago, when Professor Raymond Stevens’ research group at Scripps Research saw an opportunity to dive deeper into the science of GPCRs and the team’s foundational work was the successful initial reveal of the first human GPCR crystal structure bound to a diffusible ligand. This important discovery was followed by the characterization of other human GPCRs and coincided with over two decades of innovation in generating membrane protein structures with a focus on GPCRs to establish Structure Therapeutics.
While Structure can use its technology to develop drugs across a wide variety of GPCR targets, its initial programs focus on clinically validated targets to minimize biological risks. In addition, Structure believes that by offering small molecule drugs against targets where there are biologics administered as an injection, one can offer patients a more convenient alternative for much-needed medicines that can be dosed orally and cost less.
In only a few years from its founding in 2016, Structure Therapeutics successfully advanced two small molecule programs into clinical trials (GSBR-1290 and ANPA-0073) and one more in IND-enabling studies (LTSE-2578). The company raised $198 million privately before becoming one of biotech’s first initial public offerings in 2023. In addition, their lead program, the oral small molecule GLP-1 selective receptor agonist, has the potential to revolutionize the multiple billion-dollar obesity and diabetes market that is currently dominated by injectable and oral peptide drugs. F-Prime and Eight Roads had the foresight to know that GLP-1 was an increasingly important drug class.
The Structure Solution: Built for speed and efficiency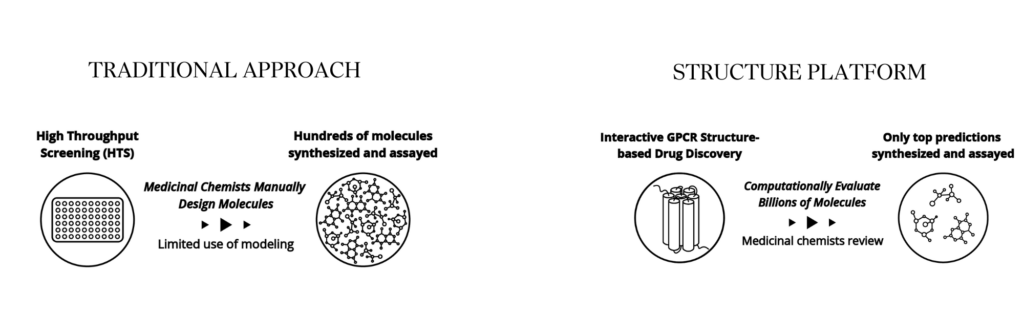
At the end of September, Structure announced positive results from the Phase 1b multiple ascending dose (MAD) study of its highly selective oral GLP-1 receptor agonist, GSBR-1290, in healthy overweight, or obese individuals. In the 28-day study, GSBR-1290 demonstrated significant weight loss supporting once-daily (QD) dosing and an encouraging safety and tolerability profile.

F-Prime acts locally to have an impact globally
At F-Prime, we help advance entirely novel technology platforms and therapeutic modalities by recognizing and furthering scientific breakthroughs. Structure was an attractive investment for F-Prime and Eight Roads from the very beginning and underscores how small molecule drug design is being transformed thanks to the explosion of cryo-EM and advances in computational chemistry.
The company created a global footprint early on, instituting its headquarters with business operations, finance, and clinical development in South San Francisco, and discovery and preclinical operations in Shanghai. This allowed the company to leverage the talent, resources, and infrastructure e.g., CROs available on both sides of the ocean. Working together with a global mindset, F-Prime’s Partners, Robert Weisskoff and Chong Xu helped to further guide the building of the company. Structure started with hiring, where F-Prime, together with Eight Roads, was key to recruiting their initial drug discovery and preclinical development leaders. The skilled group of drug developers, industry leaders, and scientific experts brought deep experience in understanding complex biological targets and mechanisms and had previously led the discovery, development, and commercialization of multiple successful drugs. The early discovery and preclinical efforts originally began in Shanghai and then followed in the U.S. with clinical activities as the company expanded its global operations. In addition, they facilitated the company’s business and financing – ultimately preparing the company for its public offering.
By empowering the next generation of companies as we did with Structure, we are proactively pioneering breakthrough science, novel technologies, and innovative platforms capable of addressing significant unmet medical needs.
“F-Prime shares our commitment to developing differentiated small molecule therapies to overcome the limitations of biologics and peptide therapies and provide better treatment options for patients everywhere.”
—Raymond Stevens, PhD, Founder and CEO of Structure Therapeutics
Shinobi Therapeutics
Shinobi Therapeutics is a biotechnology company developing a new class of off-the-shelf immune evasive iPSC-derived cell therapies. Based on the research of scientific co-founders Shin Kaneko, M.D., Ph.D., at Kyoto University and Tobias Deuse, M.D., at University of California, San Francisco, Shinobi has created a new allogeneic CD8αβ iPS-T cell platform that demonstrates comprehensive immune evasion from all arms of the immune system.
Behind the Breakthrough: Q&A with Lyn Baranowski, Avalyn Pharma
“It was really striking to me how little research and development was going on despite how serious these diseases are. Many of them are deadly, and patients have very few treatment options. I thought that there was a lot of good that I could do personally to try to bring some new treatments to market.”
There are more than 200 types of Interstitial Lung Diseases (ILD), and the most severe cases, which include Idiopathic Pulmonary Fibrosis (IPF) and Progressive Pulmonary Fibrosis (PPF), have a patient survival time closer to 3-5 years from diagnosis without a lung transplant. The disease severity is what sets the life expectancy apart for IPF and PPF and currently approved therapeutics for these respiratory illnesses can slow disease progression but carry significant side effects that limit their use. Baranowski saw an opportunity to address a large unmet need and eventually made her way to Avalyn Pharma, which was built on the mission to develop inhalable medicines that minimize systemic exposure and are more precisely targeted to the lungs. She feels she can have the most impact serving in roles at smaller companies, where “you can really see and feel the outcome of your day and how you spend time in terms of driving the company forward.”
On the heels of Avalyn’s tremendous success with an oversubscribed $175 million Series C financing in September 2023, Baranowski shares more about her background, leading and advancing Avalyn, and insights into her leadership style.
What motivated you to join Avalyn Pharma?
My career began at Novartis, where I primarily focused on the commercial, financial, and strategic aspects of the business. It was there that I was exposed to the significant unmet needs in lung diseases, particularly in rare conditions like pulmonary fibrosis, cystic fibrosis, and pulmonary hypertension. I observed the lack of research and development in these areas and recognized the severity of these diseases, often with limited treatment options.
Driven by a desire to make a positive impact, I left Novartis to work at a New York family office involved in venture capital-style investing. My biggest investment in those years was in Pearl Therapeutics, a company working on combination therapies for asthma and COPD. This experience provided valuable insights into inhalation drug delivery.
Over the years, I discovered my passion for working in smaller companies, building teams, drug pipelines, and making a difference. Despite limited resources, I found it rewarding to be entrepreneurial and witness the tangible impact of our efforts. I was heavily involved in the respiratory therapeutic area which is why my passion for addressing rare lung diseases continues to grow and I’m excited about the potential to make a positive impact in this field.
What differentiates Avalyn from other IPF and ILD players in the industry?
Most lung-focused companies still prioritize systemic drug delivery and in the case of IPF, inhaled delivery is a distinctive approach. At Avalyn, we focus on pulmonary fibrosis, a deadly disease that falls under the umbrella of interstitial lung diseases, and despite its severity, there are only two approved drugs for this condition, pirfenidone and nintedanib, both of which are administered orally. These oral medications are often poorly tolerated, leading to a high rate of discontinuation, with only 30% of patients in the U.S. taking them.
Our therapeutic focus is to improve treatment by delivering these drugs through inhalation. This approach enhances drug concentration in the lungs, improving clinical efficacy, while reducing systemic exposure, resulting in better tolerability. Inhaled delivery is itself incredibly challenging and complicated, so you really have to think about the way to deliver the medicine, how often you deliver the medicine, what the formulation is, and what the device is. It has the potential to make effective drugs more accessible and tolerable for patients, potentially extending their lives and improving their overall health. This shift in perspective is a learning curve for our field, and we hope the market will follow suit, positioning us for future success.
Avalyn’s executive leadership team and board of directors is 50% women. What’s that like, and how does it compare to the industry as a whole?
Are there any companies in our industry whose board is half women? I don’t think so. I’m incredibly proud of that. We’ve also just hired a bunch of fabulous women who aren’t necessarily on our leadership team but are VP-level. I care about hiring people who care about their jobs and patients, and I think that’s again, the right way to align as a team and be motivated.
What advice would you give to the next generation of female leaders?
Do not shy away from being empathetic. As a woman, it is important to remain committed to embracing this part of our identity. I believe in being authentic and accept that it’s perfectly fine for me to get a little emotional when a patient shares their story with us. Making a difference for patients is what drives me and I’m often touched by the stories they share with us about what living with their disease is like. It’s a natural and genuine response, and I’m proud of it.
What’s been the most rewarding part since joining Avalyn in 2022?
I love telling the story of the company, so it’s a lot of fun for me to sit down with doctors, investors, analysts, or people we’ve recently hired at Avalyn and tell them what we’re up to. It’s empowering to watch people go from not fully understanding to a moment of excitement and realization of the impact we can have.
We also just completed a $175 million financing at the end of September which feels like a huge accomplishment, especially in today’s market, and has enabled me to start hiring great people. Whether it is people that I’ve worked with before or have always wanted to work with, it feels great to now have an incredible team that we’re building and together, we’re going to do a lot of good for a lot of patients. At the end of the day, 95% of the fun I have at work has to do with the people I work with and how much I enjoy working with them.
What’s one lesson you’ve learned so far as CEO?
Don’t be afraid to be passionate and be yourself. I’m genuinely passionate about what I’m working on, and I know it translates to my peers because I’ve been told it’s infectious. If you can do that and lean into what you’re good at, then people respond to it. And, if you don’t know the answer, that’s okay. Ask your colleagues for help, advice, and feedback.
How has F-Prime been supportive during your tenure as CEO?
When I started at Avalyn a year ago, F-Prime’s Ketan Patel and Brian Yordy were invaluable and provided me with essential context regarding our historical data and the market’s overarching trends. They both possess a unique perspective when interpreting data, which enriched my own understanding and enhanced my capabilities.
How would you describe your leadership style?
I am very much a lead by example kind of person. I love talking things through and collaborating and getting people’s input. If I have a strong opinion, I’ll tell you, but often I like to hear from the team, and I think people like to be heard. At Avalyn, we’re all about integrity and keeping the patients that we’re trying to serve at the center of our focus. To me, that’s the right thing to do, but it is also the right thing for our business. If we can make decisions about how to develop our drugs with patients in mind, it means we bring drugs to market that are going to be able to impact their lives positively.
At the end of the day, we all work in this industry because we care about making a difference in patients’ lives and if we can keep that as our driver and our mission, it’s a great way to remind people of why we all wake up in the morning and what we’re trying to do together.
What makes you most hopeful about Avalyn’s future on the heels of a successful series C fundraise?
We’re in the process of launching a significant clinical trial for inhaled pirfenidone (AP01), and our focus is on diligently executing the study startup. This trial is set to be a large, well-controlled global effort, representing a crucial step forward for inhaled pirfenidone’s path to market, which is a very exciting development.
The capital we just raised also allows us to do the next stage of work with AP02 (inhaled nintedanib), which is the second lead clinical asset, and continue our efforts on the preclinical work for the fixed-dose combo of the two together. By transitioning from one mechanism to two, this combination therapy has the potential to revolutionize our field.
A lot of exciting initiatives are going on and it is my priority to build the company, grow the team, and do all the important work to set us up for success for the next stage.
Subscribe to our newsletter to get the latest updates on how our portfolio companies continue to lead the way in their respective fields.
T-Therapeutics
T-Therapeutics is a next-generation T cell receptor (TCR) company spun off from the University of Cambridge. The company was created to harness the power of T cell biology, evolved over millions of years, to create safe and effective treatments for many cancers and autoimmune diseases.